- Home
- Health Center
- Health Info
- Covid-19 Tests: RT-PCR vs RTK Antigen Test
Test
Covid-19 Tests: RT-PCR vs RTK Antigen Test

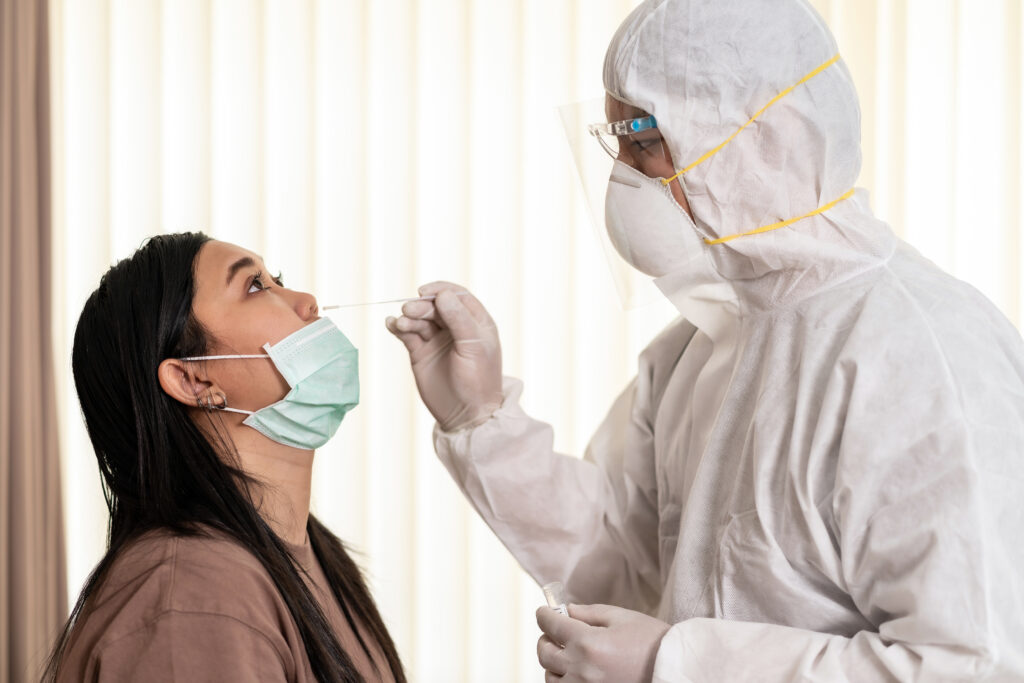
Most of us know that a swab test is carried out to test active Covid-19 virus infection during this pandemic. Specimens will be collected via a 6-inch specialised swab stick that is inserted into our nose and/or throat, and sent for further diagnostic testing. However, do you know what are the common diagnostic tests available in Malaysia? Let us break it down for you!

RT-PCR (Reverse Transcription-Polymerase Chain Reaction)
RT-PCR test is performed to identify genetic material (RNA) from the virus. Most RT-PCR specimens will be taken via nasal or throat swab, and most recently, saliva testing may be available in public settings. 1,2
Accuracy. This test is recognized as the gold standard by the Ministry of Health (MoH) for diagnosis of active Covid-19 infection due to its higher sensitivity (≥ 90%) and specificity (≥ 97%) rate among other diagnostic tests. 1,6
*Test sensitivity is the ability of a test to correctly identify persons with infection, whereas test specificity is the ability to correctly rule out infection.
Limitations. However, since this test requires a more sophisticated process and equipment, it comes with a higher price per test and a longer turnaround time (1 – 3 days) to acquire the result. 3,5,6
Results. The positive test result indicates most likely that you have been infected with the Covid-19 virus infection. This positive result should not be repeated within 90 days since infected individuals may still have fragments of virus genetic material even after the risk of transmission has reduced to minimal. 1,6
RTK-Ag Test (Rapid Test Kit- Antigen Test)
RTK-Ag test is carried out to detect viral proteins related to the Covid-19 virus to determine active infection. 1,2
When is it used? This method is considered by MoH as an alternative to RT-PCR test in certain circumstances, which can result in a shorter time frame (approximately 15 – 30 mins) to allow rapid identification of infected people, thus avoiding further virus outbreak in a setting. It also comes at a lower price per test compared to RT-PCR. Typically, the test would need a specimen taken via nasal or throat swab, and saliva samples.1,2,7
Accuracy & limitations. However, the disadvantage of this test is that it has a lower sensitivity rate (60 – 80%) than RT-PCR, indicating a higher risk of false-negative results, especially among asymptomatic people. This may need a confirmatory test using RT-PCR as a gold standard.3,5,7
Take Away
To sum it all up, both RT-PCR and RTK-Ag tests are effective to detect current Covid-19 infection. If you have any related symptoms or are in close contact with positive Covid-19 individuals, please get tested! However remember that testing negative today does not mean you won’t contract the virus tomorrow. It’s important to continue to practice physical distancing and hand hygiene.
If you have further inquiries about the tests, you may consult our pharmacist at any CARiNG Pharmacy stores or message us on our Facebook page. Until then, stay safe and healthy everyone!
Reference:
- COVID-19 Malaysia Updates. Ministry of Health (MOH) of Malaysia. (Web accessed June 2021). Web link: http://covid-19.moh.gov.my/faqsop/faq-covid-19-kkm
- Coronavirus Disease 2019 Testing Basics. U.S. Food and Drug Administration (FDA). (Web accessed June 2021). Web link: https://www.fda.gov/consumers/consumer-updates/coronavirus-disease-2019-testing-basics
- Laboratory Diagnosis of COVID-19: Current Issues and Challenges. Tang Y, Schmitz J, Persing D, Stratton C. Journal of Clinical Microbiology. 2020;58(6).
- ESC Guidance for the Diagnosis and Management of CV Disease during the COVID-19 Pandemic. European Society of Cardiology (ESC). (Web accessed June 2021). Web link: https://www.escardio.org/Education/COVID-19-and-Cardiology/ESC-COVID-19-Guidance
- Laboratory diagnosis of COVID-19.Goudouris ES. Jornal de Pediatria (Rio J). 2021;97(1):7-12.
- Nucleic Acid Amplification Tests (NAATs). Centers for Disease Control and Prevention (CDC). (Web accessed June 2021). Web link: https://www.cdc.gov/coronavirus/2019-ncov/lab/naats.html
- Interim Guidance for Antigen Testing for SARS-CoV-2. Centers for Disease Control and Prevention (CDC). (Web accessed June 2021). Web link: https://www.cdc.gov/coronavirus/2019-ncov/lab/resources/antigen-tests-guidelines.html
- Rough estimate. Actual prices may defer. Information retrieved from websites of multiple healthcare centres, private hospitals and newspaper articles
Latest Health Info
The Role of Vitamin D in a Healthy Pregnancy
Vitamin D is an important nutrient that supports both the mother’s health and the baby’s development during pregnancy. It helps ...
Building Strong Bones for Growing Kids
Strong bones are like a child’s internal scaffolding: they support growing bodies, protect organs, and allow children to run, jump, ...
Nutrition in Pregnancy: Fueling Two Lives for a Healthy Future
Pregnancy is a transformative experience that requires extra care when it comes to nutrition. The development of the baby depends ...



